Predicting CO2 levels
Just a bit of math for alarmists
The CO2 levels in the atmosphere measured as parts per million by volume is accurately modeled by the equation:
CO2 = 59.25 x e^(.016 (t-1959) + 255.48
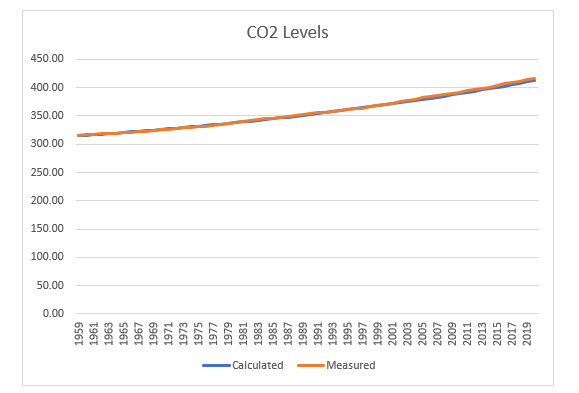
Here are the calculated levels using that deterministic function compared with the measurements reported by the climatologists at Mauna Loa.
The fit is so good the lines overlap one another when the data are charted.
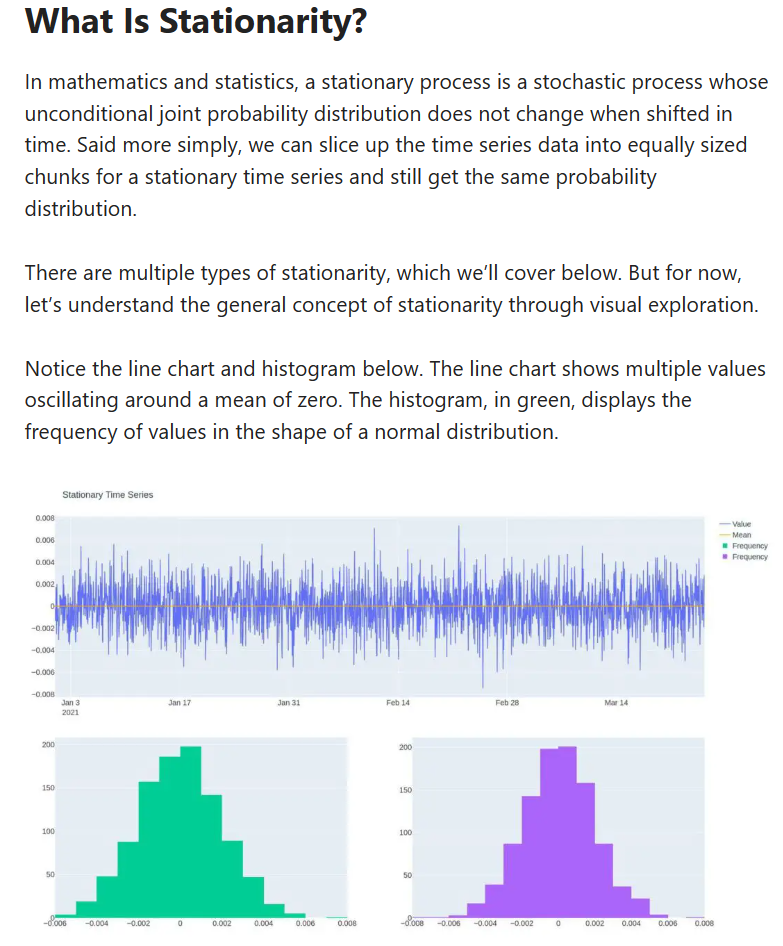
From a mathematical point of view, the function set out above comprises a “stationary process”. That is impossible if CO2 levels are caused by mankind’s fossil fuel emissions - the function presented accurately predicts CO2 levels in the atmosphere independent of fossil fuel emissions which do not comprise a term of the function. All changes are simply a function of time. In the case of changes in climate, the appropriate time frame is geological time, that is, millions of years. The tiny changes in the past century or two are rounding errors in a larger process and not materially affected by human activity.
The concept of stationarity is important in this context.
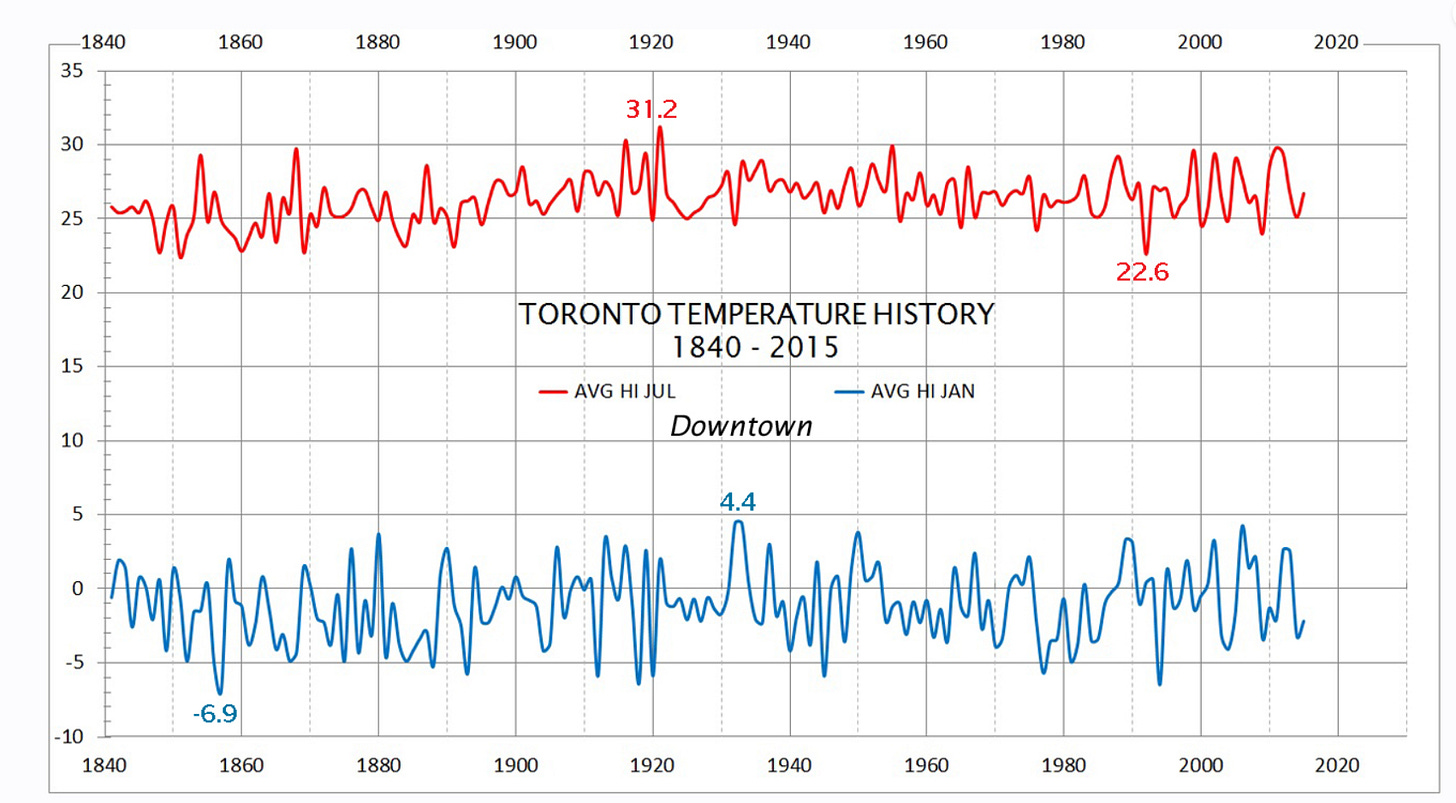
Note the similarity between the line chart showing random values in a Gaussian distribution around a mean of zero and and a temperature chart for any measuring station on Earth. Here is one for Toronto since 1840 with the noise removed.
No amount of “climate science” can change reality. Toronto temperatures are not rising over the past 180 years. Nor are “global average” temperatures - a nonsensical concept incapable of accurate measurement, making it perfect for those who want to promote alarmist rhetoric.
Canada’s Liberal government is squandering billions of dollars that could be used to mitigate homelessness and poverty and fund pensions by both attacking the fossil fuel industry and enacting costly “climate policies” that pander to the World Economic Forum but have no ability whatsoever to alter the course of nature. We need to toss Trudeau out of office.

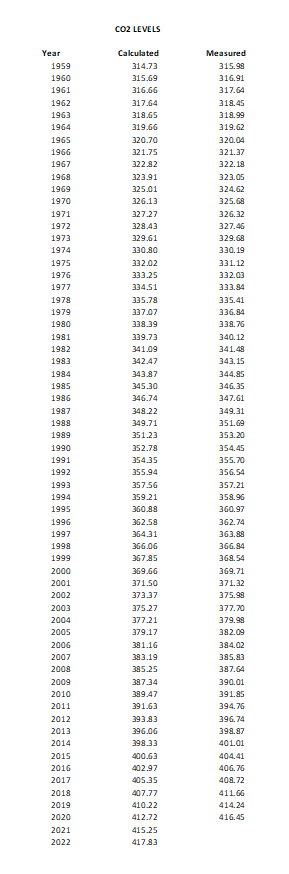
Probably they will say that the values in 1840 are not accurately measured, bla bla bla.
It´s always a difficult subject, but I like your approach!
Cheers.